Spring dead spot (SDS) is the most damaging disease of bermudagrass turf. This disease is caused by three, closely related root pathogens, but Ophiosphaerella korrae is the most common causal agent in the mid-Atlantic. In Australia, Ophiosphaerella narmari is the most common agent, and in parts of the semi-arid western states Ophiosphaerella herpotricha is most common. The fungal cause [i.e., the pathogen(s); Ophiosphaerella spp.] of spring dead spot was first reported Dr. A.M. Smith, a Dept. of Agriculture Researcher in New South Wales, Australia in 1971.

Spring dead spot appearing mid-April, 2022 in a bermudagras fairway in Georgetown DE.
Discovery and description of the pathogen went unknown in the USA, until a graduate student at the University of Maryland, documented that O. korrae was the cause of the disease in the mid-Atlantic in 1988. Joelle Crahay was among the many special students who appeared in my office to say they wanted to study plant pathology. Joelle Crahay had a BS from The University of Liege in Belgium, where she was trained to be a pre-natal nurse. She ended up in Maryland with her husband, who was a Shock Trauma physician at the University of Maryland medical school in Baltimore. Joelle was a Shock Trauma nurse, who experienced some of the most horrible medicals traumas imaginable. One incident in the ER turned her life away from medicine, and she ended up with me working on spring dead spot (SDS) of bermudagrass in College Park.
At that time, SDS was recognized as the most destructive disease of bermudagrass, yet nobody in the US had a clue as to the cause. With information published by A.M. Smith in 1971, we knew we should be searching for a darkly-pigmented fungus that attacked roots and stolons. Using old fashioned lab techniques, she was able to produce in the lab the sexual fruiting bodies (black-flask-shaped structures called pseudothecia in which long-needle shaped spores are produced) of the pathogen on naturally infected bermudagrass roots. The research began with trying to isolate the fungus from infected tissues. The problem was that even if we isolated the right fungus, we had an uphill climb to proving we had the right pathogen via the time-honored proof provided by spores (spores are not produced on agar in a petri dish). The sure-fired way to short-cut a massive job, was to try and rear the fruiting bodies on infected tissues. We need to find the spores. Joelle selected obviously infected root masses; cleaned them with a disinfectant, and set them-up in sterile test tubes wrapped in sterile gauze and distilled water. Large numbers were prepared and racks of test tubes were placed in various locations in the lab. After about 100 days (maybe longer), Joelle wanted to throw everything away. So, we sat down under a microscope, opened each test tube and looked. Bingo!-The winner came from test tubes set on a bench next to a window. Small numbers of the black flask-shaped pseudothecia (i.e., fruiting bodies in which spore would be borne) were present on roots; she then isolated the fungus from spores on a sterile agar medium. Measuring numerous spores, we knew she had found O. korrae from a description of the genus (then named Leptosphaeria –changed to Ophiosphaerella –fungal taxonomy is complicated), publish by Walker and Smith in Australia in 1972.

Crahay incubated naturally infected bermudagrass roots for more than 100 days before fruting bodies were produced by the pathogen..
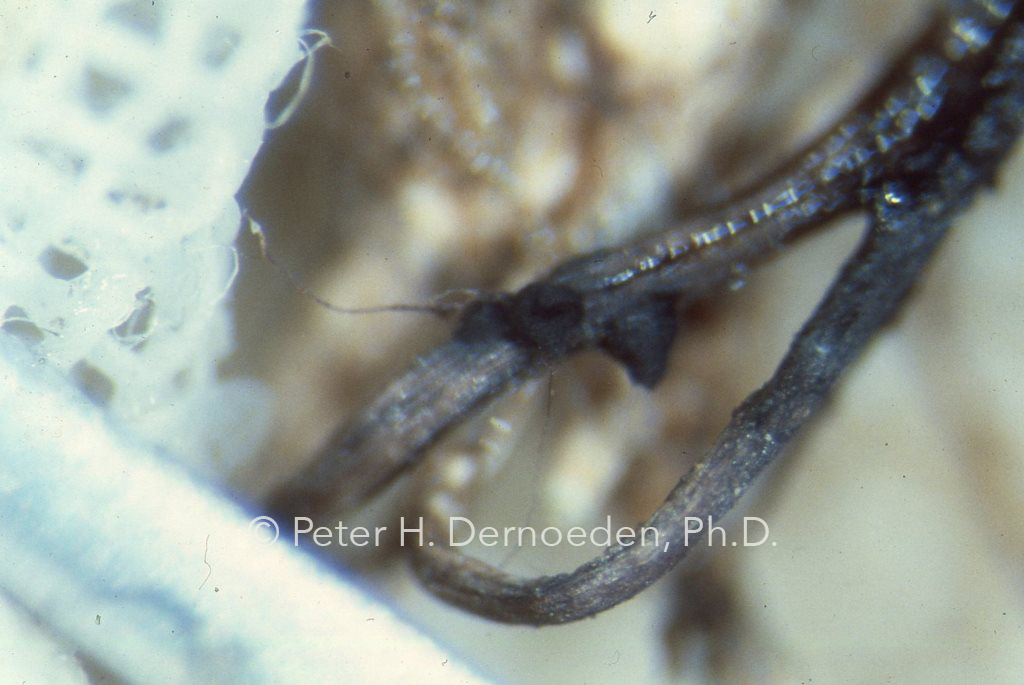
Bingo- the first black-flask-shaped pseudothecia of O. korrae on infected bermudagrass roots in the U.S.A. came from a UM lab.
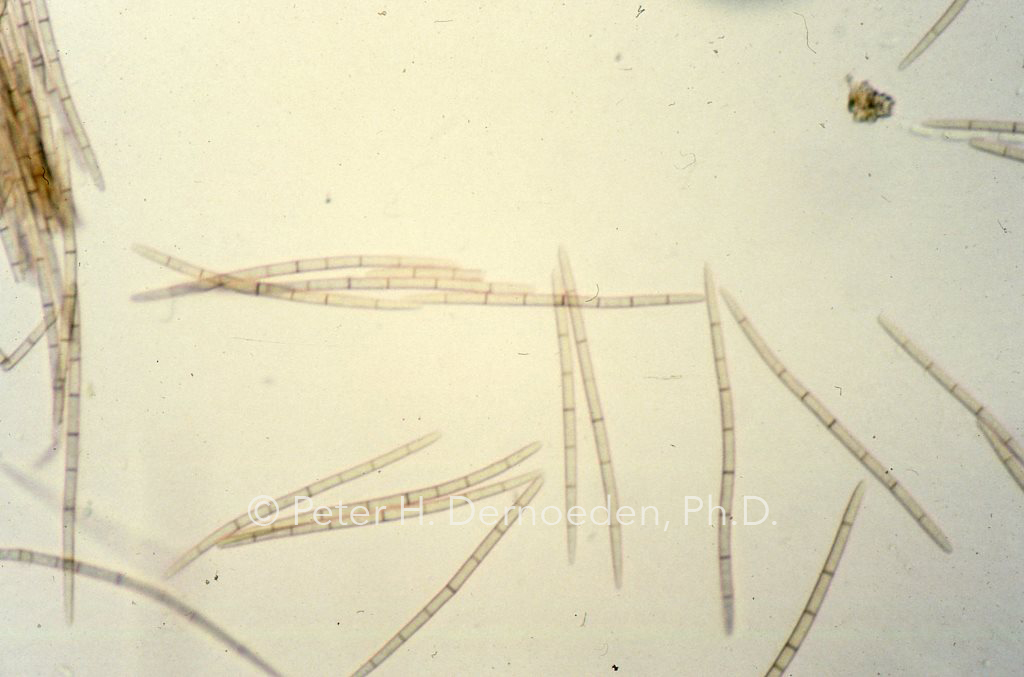
Long, brown needle-shaped spores from pseudothecia proved for the first time in the US that O.korrae an incitant of SDS.
Subsequently, Joelle conducted inoculation studies at different temperatures to not only prove pathogenicity of our fungus (what is called Koch’s Postulates), but also described the influence of temperature on infection (see below). Furthermore, she conducted field trials that showed that ammonium sulfate was the best N-source to help speed recovery of living plants into dead patches. University’s today place too much emphasis on molecular biology today, which largely is cookbook/boilerplate work. “Muddy knee-field research” combined with lab work, as shown in Joelle’s research, provides the most valuable information for working turf professionals.

O. korrae's dark-colored mycelium was first isolated in the USA from ascopores at UM in 1986.
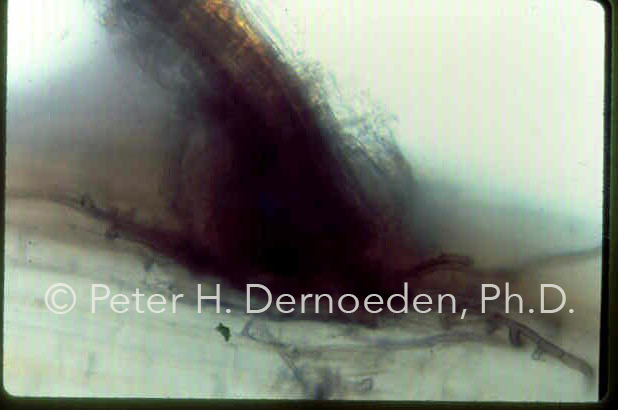
Note the darkly pigmented mycelium of O. korrae wrapping itself at the point where secondary roots develop.
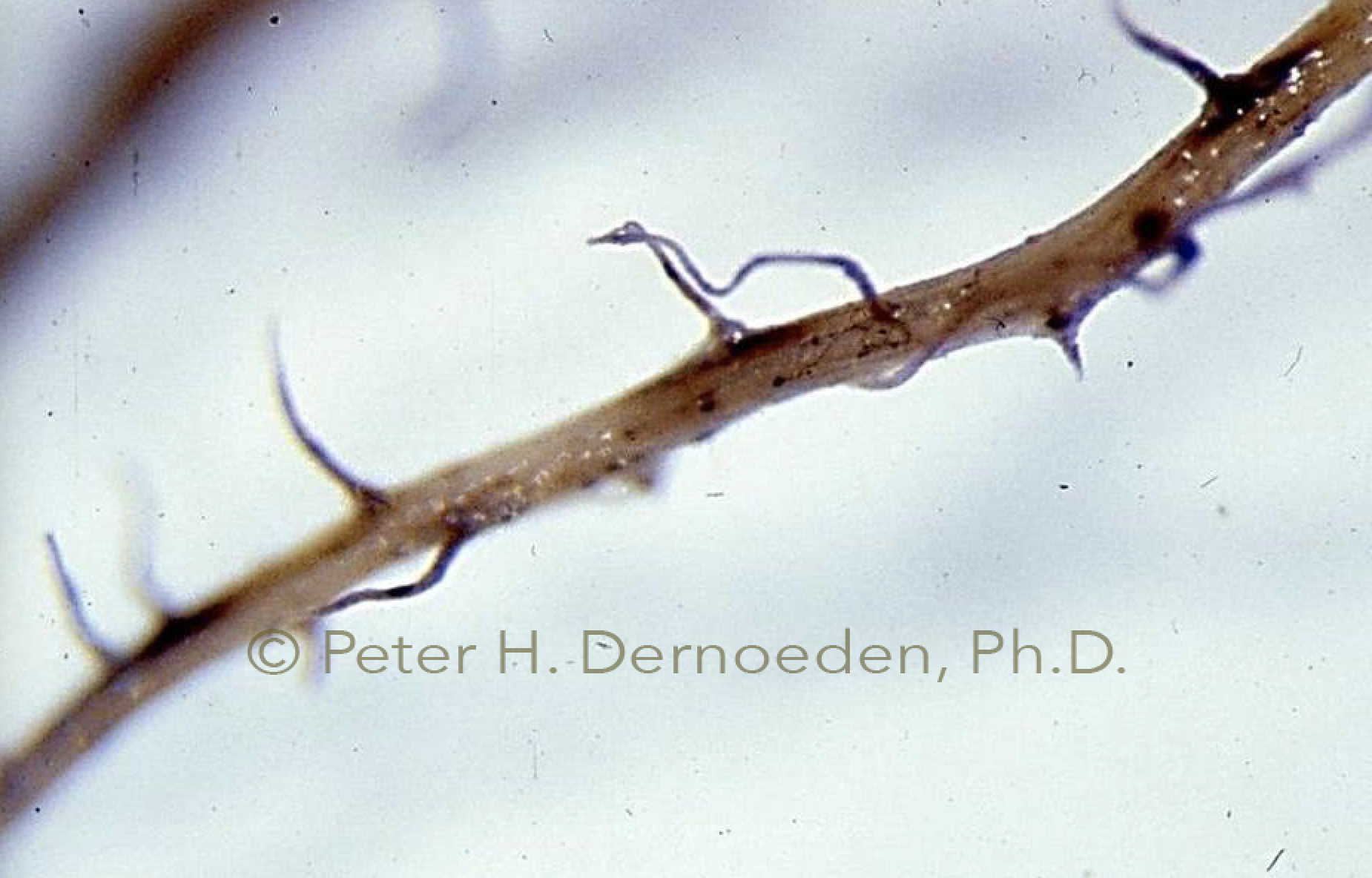
Bermudagrass primary roots are thick and wiry; note secondary roots rotted; fungus is progressing to primary roots and then onto stems.

Spring dead spot (SDS) patches in a 'Patriot' bermudagrass fairway.
Symptoms: As bermudagrass breaks dormancy in spring (about mid-to-late April in our area), circular patches of tan or brown dead turf a few inches to three feet or greater in diameter become conspicuous. Circular depressions sometimes may be seen in affected turfs prior to spring green-up. In older stands with chromic SDS, rings may develop with healthy turf in the center, indicating that the pathogen is moving outwards; much like older fairy rings. Sometimes patches coalesce, and the damage is non-uniform and appears similar to winter-kill or winter desiccation. Rhizomes (below ground stems) and stolons (above ground stems that root on soil surfaces) from nearby healthy plants eventually spread into and cover dead patches. This filling-in process is slow, a period which may last four to eight weeks. Unusually cool to cold spring and early summer weather further delays recovery.

Spring dead spot in a bermudagrass soccer field in Baltimore.

Coalescing SDS patches may give turf a winter kill appearance, June 2015.
General Information: The SDS pathogen(s) attack in soil on roots, stolons and rhizomes; they are not found on foliage. The intensity of SDS varies greatly from year-to-year, and it is not possible to predict those years when it will be severe. Indeed, turf pathologists are baffled by the unpredictable nature of SDS outbreaks. The disease generally is expected to be more common and severe during springs following cold and wet winters, but severe outbreaks of SDS have followed relatively mild winters. Thus, the intensity of epidemics is due to imperfectly understood environmental and soil conditions. This spring, however, we now recognize that we have a severe outbreak on our hands.
Temperature and SDS. Joelle Crahay showed that root infection by O. korrae occurs when soil temperatures range from about 50˚F, but most root injury occurs between 55 to 65˚F. These soil temperatures coincide with the time bermudagrass enters dormancy in late autumn. Temperatures below 50˚F restrict growth of the pathogen and thus little injury would be expected to occur overwinter. The Maryland researcher also showed that O. korrae was unable to damage roots when inoculated plants were maintained at about 75˚F, accounting for why SDS is not a major concern in regions where bermudagrass does not go completely dormant in winter.

Crahay inoculated bermudagrass and incubated plants under various temperatures to reveal that max. injury occurs in autumn when soil temps are in the high 50'AF.
Management practices that help reduce thatch (e.g., vertical cutting and coring) are very important in an SDS management program. Compaction is a growing problem on golf courses given the high increase in play during the last two (COVD) years. The aforementioned practices are best performed in early summer after 100% green-up and when daily highs exceed 80˚F to promote growth and recovery. Potassium inputs also are important, despite soil tests showing sufficient K levels. I have always recommended applying nitrogen (N) and potassium (K) in a 1:1 ratio whenever using N in bermudagrass. Ammonium sulfate (applied at 1.0 lb. N/1000 ft2) and potassium chloride (applied at 1.0 lb. K/1000 ft2) applied on 14 to 21 day intervals (or lower rates more frequently) beginning at spring green-up speed recovery and help to alleviate SDS severity over time.
This type of practical management information was generated by a three year Maryland field (no way to do this in a lab) study, which assessed the ability of ammonium sulfate, ammonium nitrate, urea, sodium nitrate and other water soluble fertilizers in speeding recovery. All water soluble N sources speeded recovery versus no N, but ammonium sulfate was more rapid and provided a faster and better greening effect. The study was conducted over a 3-year period, and in the 3rd year ammonium sulfate suppressed SDS by reducing patch size, but not the number of patches. In that study, ammonium sulfate was applied at 1.0 lb. N/1000 ft2 (total N ranged from 3-4 lb/1000 ft2) on 14 to 21 day intervals beginning at spring green-up. Since the suppression effect on patch size took 3 years to develop, do not expect any immediate “magic” from an ammonium sulfate program. Extrapolating results, it appears prudent to apply ammonium sulfate at 0.5 to 0.75 lb. N/1000 ft2 every 7 to 14 days (depending on severity) to speed recovery. The suppression effect provided by ammonium sulfate (via soil acidification) may take several years to develop. Acidification alone does not reduce the number of patches, but reduces their size. Late fall N applications (within 30 days of dormancy) of high amounts of water soluble N (> 2.0 lb N/1000 ft2) have been shown to enhance SDS. It is likely, however, that modest (0.25 to 0.50 lb. N/1000 ft2) inputs of nitrogen (N) in early autumn, especially from slow release N forms, would not be expected to intensify the disease. Frequent applications of water soluble nitrogen (N) are the only known approach to speeding recovery of turf damaged by spring dead spot (SDS).

J. Crahay (blue striped jersey) applied several N-sources and found ammonium sulfate produced best results. Caroline CC, Denton; Jim McHenry supervises.
Weeds commonly invade dead SDS patches, and they compete for space and slow recovery. A rule of thumb is that bermudagrass requires 100 days without weed competition to fully attain optimum density in the mid-Atlantic. Hence, it is very important to eliminate weeds as soon as possible. Most preemergence herbicides targeting annual grass weeds in spring could affect rooting of stolons, especially on close cut fairways and athletic fields. RonStar (oxadiazon) is preferred since it is so safe on rooting that it can be used overtop bermudagrass sprigs. However, only the granular form of RonStar is safe to use after bermudagrass begins to green-up. RonStar also is very expensive, but any preemergence herbicide is better than none. In the below photo you can see overseeded perennial ryegrass in dead SDS patches will prevent complete recovery via its weed-like competition. Simply stated –get the weeds and all other competitors out of bermudagrass ASAP.

Tee over- seeded with perenial ryegrass to mask damage. The presence of ryegrass will likely prevent total recovery. Remove all weeds and other competitors ASAP.
Irrigate SDS affected turf to prevent wilt during dry periods and maintain N applications throughout summer to encourage re-growth by stolons and rhizomes. Improve drainage in low areas where water is likely to puddle and freeze in winter. Increasing mowing height in September will allow bermudagrass to produce and store more carbohydrates in stolons and rhizomes, which should improve winter survivability and recovery from SDS. Potassium applied in late summer and autumn also can help to improve winter hardiness of bermudagrass.
Fungicides: Joelle’s research clearly showed that there is no benefit to be achieved from an early spring applications of fungicides. Obviously, most of the damage occurs in the autumn and early winter period, and by the time symptoms appear in spring –maximum damage has occurred. We are not familiar with any fungicide able to bring dead turf back to life!

Dr. Pete had great graduate students- it's time to bring back the tradition.
